

CeQur Simplicity™ is a 4-day wearable insulin patch that is clinically proven to reduce patients’ A1C and increase time-in-range1
Why Prescribe the CeQur Simplicity Wearable Insulin Patch?
Patients that use the CeQur Simplicity wearable insulin patch are more likely to adhere to their insulin regimen and achieve glycemic targets.
CeQur Simplicity is simplifying mealtime insulin delivery for T1 and T2 diabetes patients on multiple daily injections (MDIs). Our 4-day wearable insulin patch removes mealtime insulin barriers, providing a convenient, discreet, and injection-free way for people with diabetes to deliver mealtime and correction boluses.1

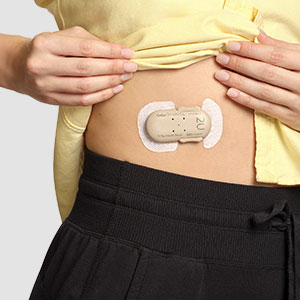
Convenient
Can be worn for up to four days1
Water-resistant3
Holds up to 200 units of rapid-acting insulin1
Easy to use and learn, <30 minutes of training2

Discreet
Can be worn and dosed under clothing
One click administers 2 units of insulin
Thin (<4 stacked quarters), compact (65 x 36 mm), and lightweight (10 gm)
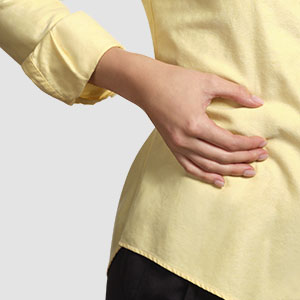
Injection-Free
Less pain encourages adherence1
Clinically shown to reduce A1C, increase TIR1,4
One patch replaces up to twelve mealtime injections
Small, flexible cannula for painless insulin delivery1

CeQur Simplicity is Easy to Teach and Use
Getting patients started with CeQur Simplicity is easy. Most patients need <30 minutes of training and feel fully confident applying the patch after just 1-3 times. Healthcare professionals also found that training patients to use the patch was easy.2
Patients follow these simple steps to begin using the CeQur Simplicity 4-day wearable insulin patch*
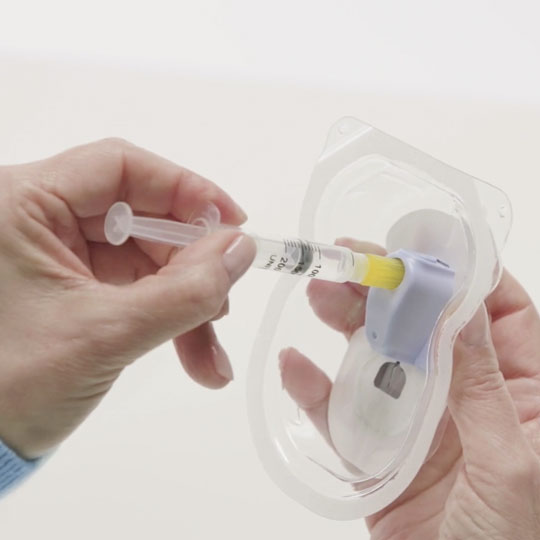
Fill the patch with a 4-day supply of insulin. CeQur Simplicity holds up to 200 units of rapid-acting insulin*
*Labeled for use with Humalog® U-100 and Novolog® U-100
1

Use the Inserter to apply the patch to the abdomen for up to four days of wear and injection-free dosing
2
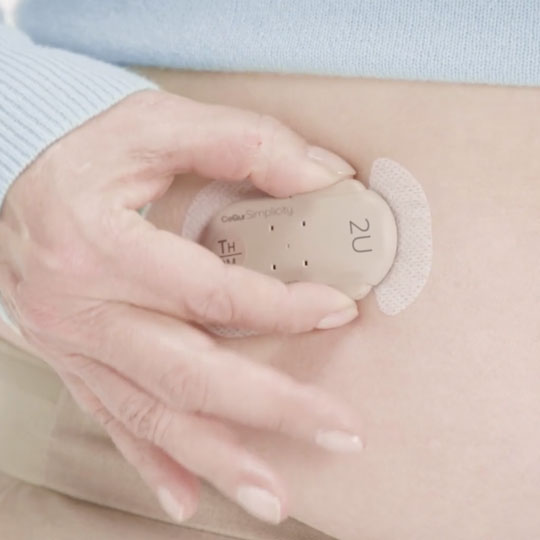
Remove the Inserter, and the patch is ready to dose.
3
The CeQur Simplicity wearable insulin patch holds up to 200 units of rapid-acting insulin administered in 2-unit increments.1


9 out of 10 prefer CeQur Simplicity over pens and syringes2

Built-in safety features ensure proper dosing and quickly build patient comfort
Two-button safety mechanism prevents accidental dosing. Buttons lock in “squeezed” position when reservoir is empty or cannula is occluded3
Audible and tactile feedback indicates 2-unit delivery3
Water-resistant to a depth of 1 meter for up to 30 minutes3
Our Wearable Insulin Patch is
Proven Reliable and Effective
CeQur Simplicity is clinically shown to improve glycemic control, which can reduce risk of long-term health complications.
Baseline
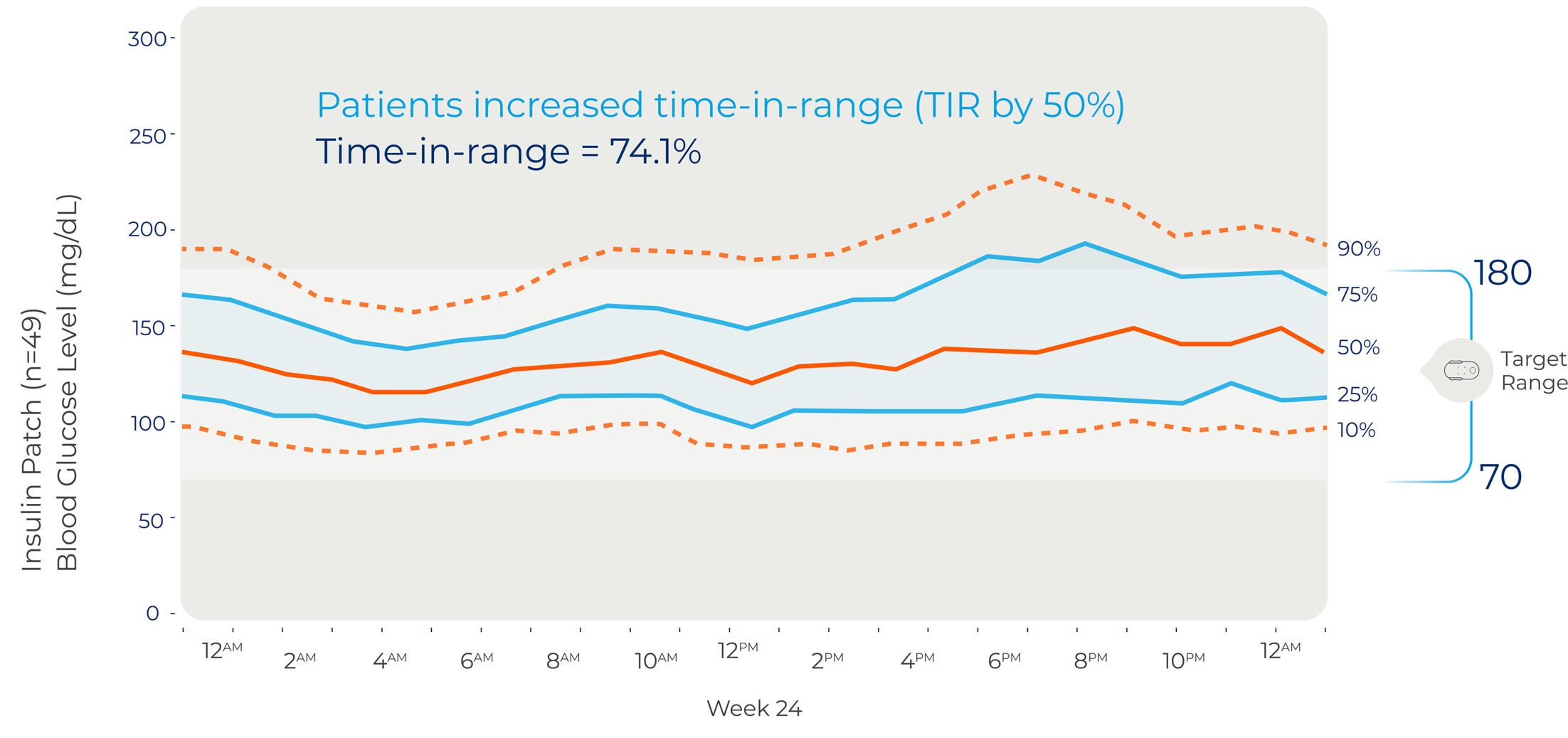
Week 24
Week 44
After 24 weeks patients decreased their A1C an average of 1.7% and increased time-in-range (TIR) by 50%1,4
Results were comparable to pens. Consistent A1C <7% supports HEDIS Comprehensive Diabetes Care measure
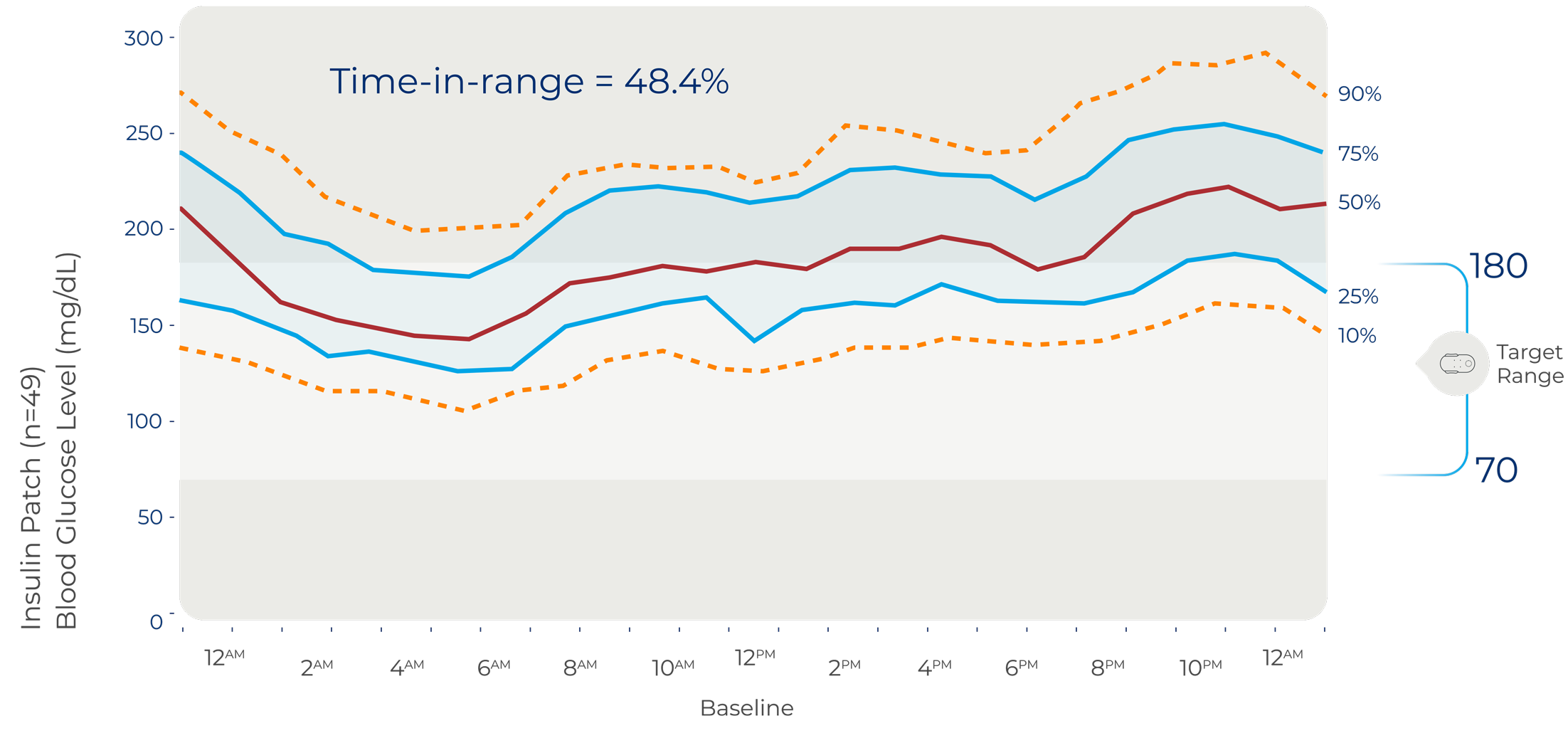
The International Consensus on Time-in-range defines clinical target for TIR > 70%, which is evidenced to be the equivalent to an A1C < 7%5
Healthcare Professionals and Patients Prefer CeQur Simplicity
Healthcare professionals prefer the patch to a pen when advancing people with diabetes from basal to basal/bolus insulin management.
We Have You and Your Patients Covered
CeQur Simplicity is covered on most commercial and Medicare Part D insurance plans as a pharmacy benefit. In plans where CeQur Simplicity is covered, most patients pay less than $35 for a 30-day supply of CeQur Simplicity. For eligible patients whose commercial insurance does not cover CeQur Simplicity or have a high copay, the CeQur Simplicity CoPay Savings Program may help. Eligible, commercially insured patients may pay less than $35 for a 30-day supply† of CeQur Simplicity and no more than $5 for an Inserter†. Learn more at CeQur Simplicity Copay Savings.
For additional questions, contact the CeQur Cares™ Team at 1-888-552-3787.
How to get patients started with the CeQur Simplicity wearable insulin patch:
- 1 Prescribe CeQur Simplicity 2U patch, 8-pack
- 2Prescribe CeQur Simplicity Inserter
- 3Prescribe a vial of rapid-acting insulin – labeled for use with Humalog® U-100 or Novolog® U-100
- 4Ensure patients schedule their training session to get started. Training must occur prior to first usage. A healthcare provider from your office or a CeQur Cares healthcare professional can train the patient. Have patients contact CeQur Cares Team at 1-888-552-3787 or cequrcare@cequr.com.
Training Checklist will help with preparation and day-of-training readiness. Download here
Patient Resources
Healthcare Professional Resources
We’re Here to Help You and Your Patients Get Started
CeQur Simplicity’s 4-day wearable insulin patch simplifies mealtime insulin management for T1 and T2 diabetes patients, reducing the need for multiple daily injections (MDIs). Our injection-free wearable insulin patch can improve A1C and time-in-range (TIR) for better health outcomes.
Call Us
1-888-55-CEQUR (1-888-552-3787)
Email Us
Get Your Patients Started Today
We respect the importance of your privacy and understand your health is a very personal and sensitive subject. CeQur Simplicity wants you to understand how it will use the information provided by you on this registration page. By clicking the “Submit” button above, you are indicating that CeQur Simplicity or its partners may use your information for program improvements, quality monitoring, data and analytics for commercial purposes, and future marketing communications. You also understand that you may opt out from receiving any future communications from CeQur Simplicity or its partners by clicking the “unsubscribe” link within any email you receive, or by calling 1-888-552-3787.
To better understand how CeQur Simplicity values your privacy, please see our Privacy Policy.
References
Humalog® is a registered trademark of Eli Lilly and Company. NovoLog® is a registered trademark of Novo Nordisk A/S.
† For eligible, commercially insured patients. These offers are not valid for use by cash-paying patients or patients enrolled in Medicare, Medicaid, and TRICARE or other federal or state programs. Not valid where prohibited by law. Offers are subject to quantity limitations and maximum benefit amounts per fill apply. CeQur® reserves the right to rescind, revoke, or amend this offer without notice.